
Restoring Lung Function With Nanotechnology
OUR MISSION
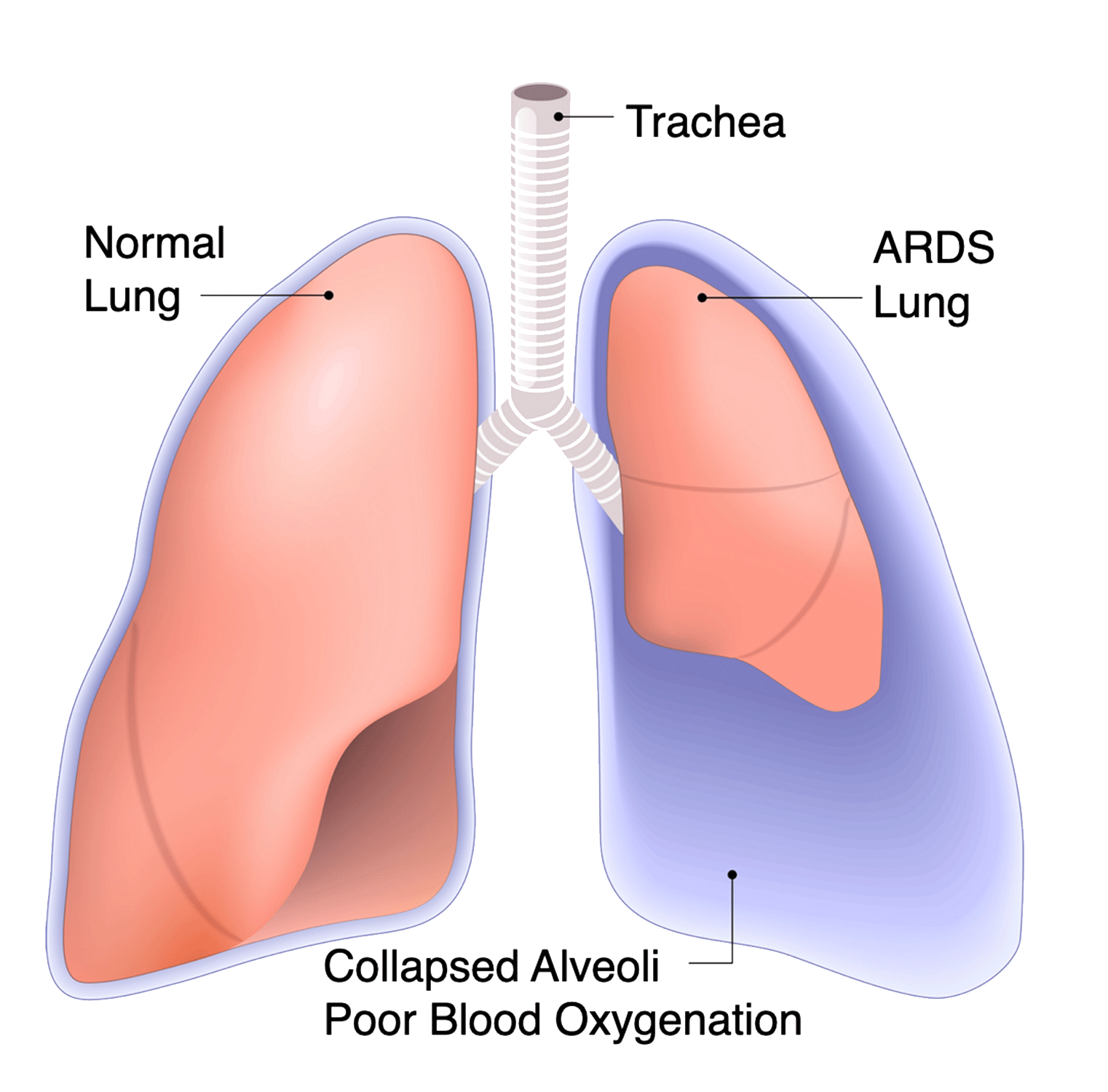
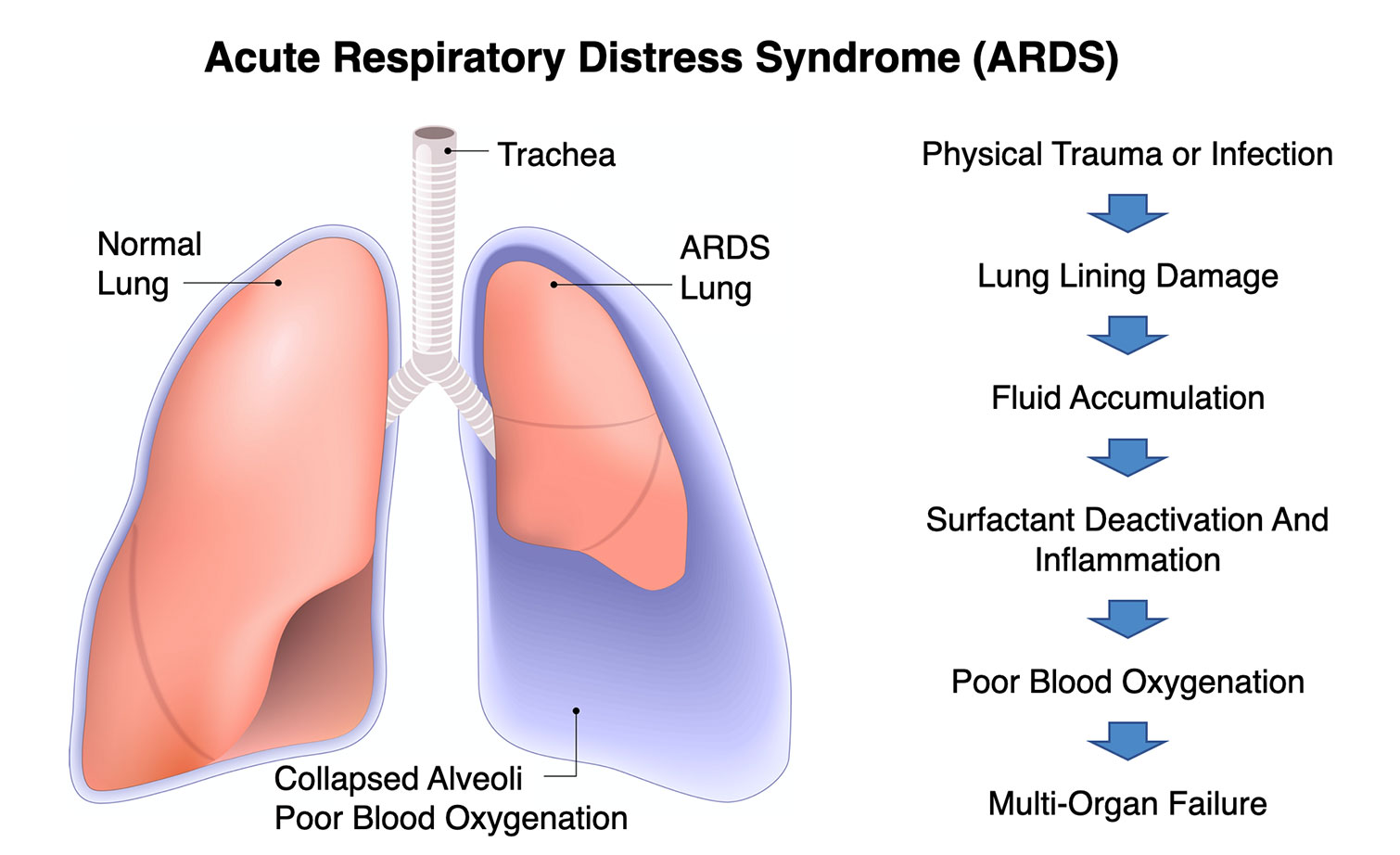
Lung Therapy For ARDS
Acute respiratory distress syndrome (ARDS) is a life-threatening lung injury that allows fluid to leak into the lungs. Breathing becomes difficult and oxygen cannot get into the body. Most people who get ARDS are already at the hospital for trauma or illness.
Our mission is to providing lung restoration to babies suffering
from ARDS by using new proprietary methods
OUR SOLUTION
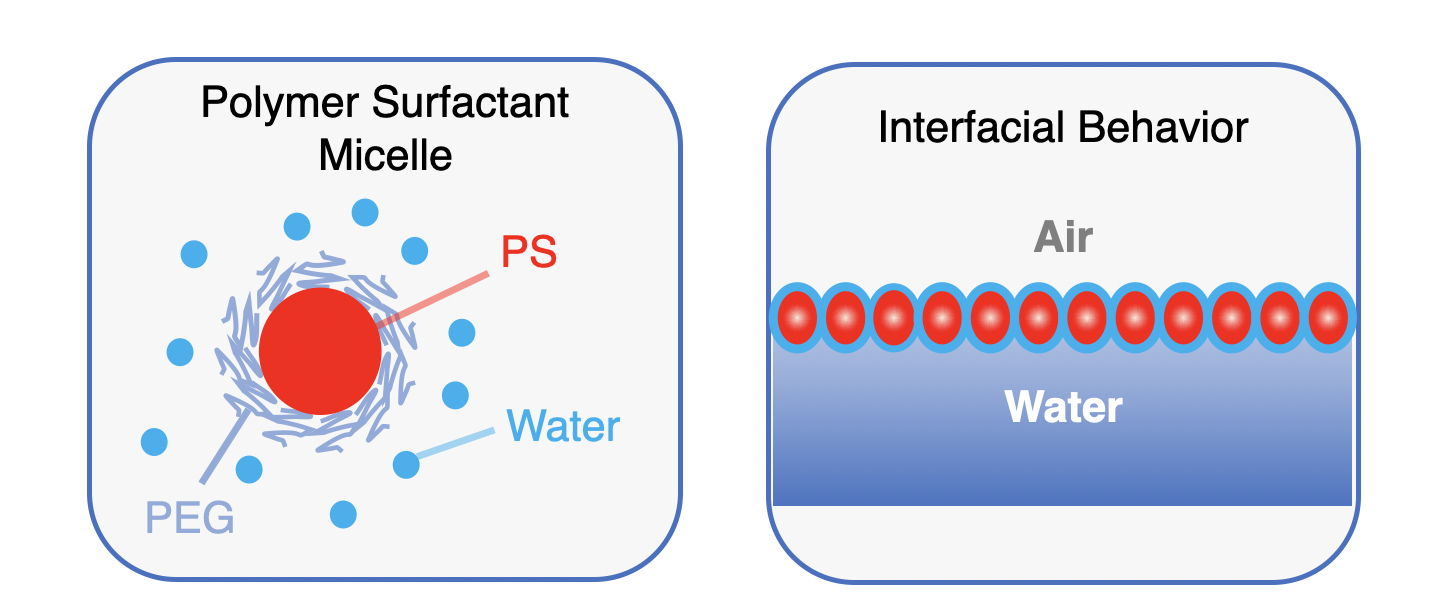
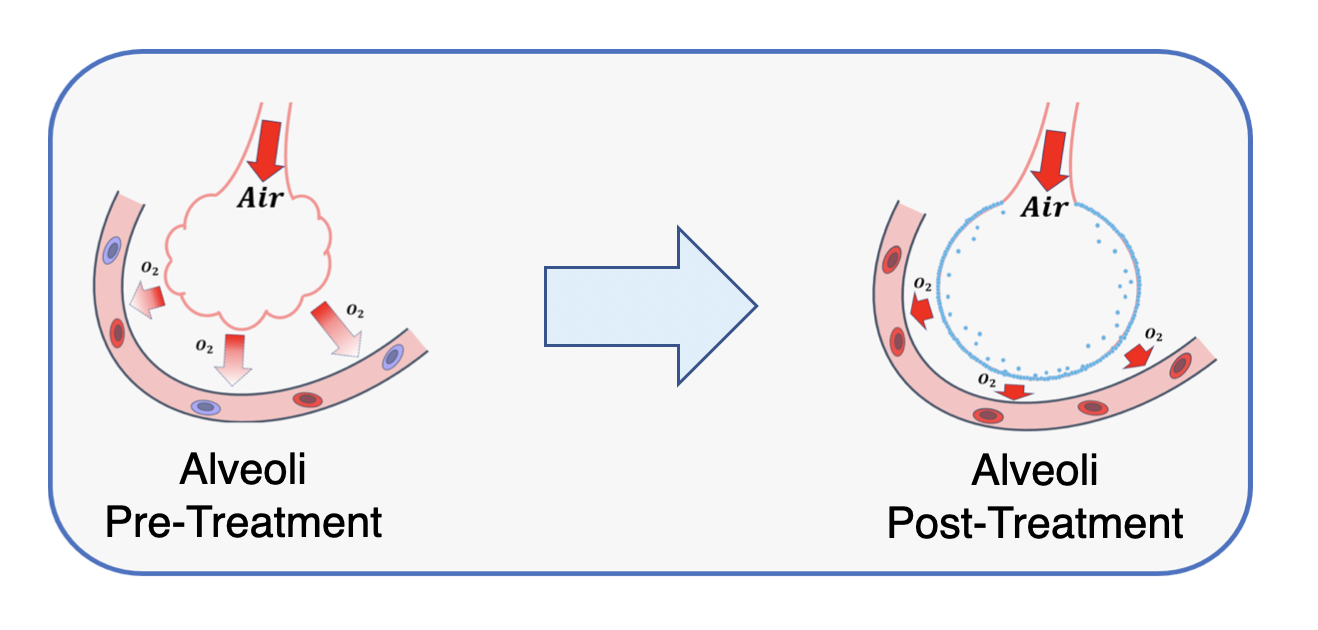
A First-In-Kind, Polymer Lung Surfactant (PLS)
- Low complexity synthesis and formulation
- Interface affinity exceeds competing agents
- Reduces alveolar surface tension and fluid accumulation
OUR JOURNEY
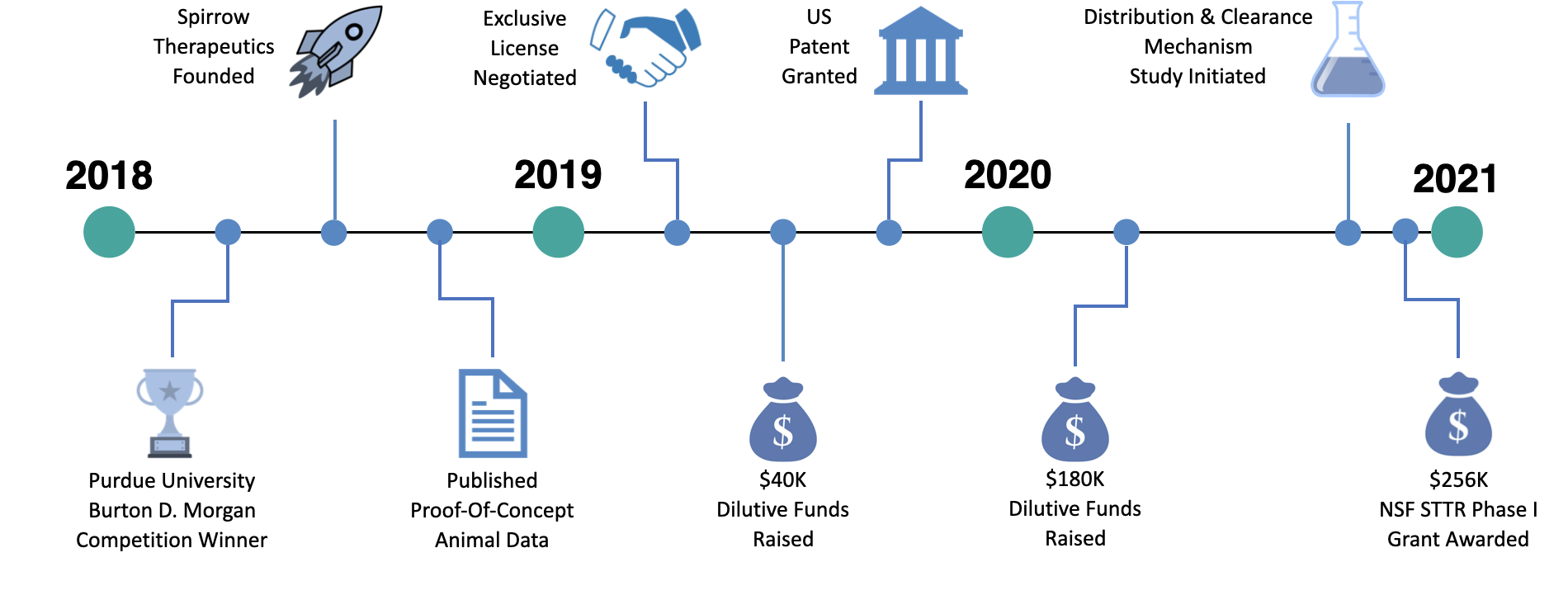
MILESTONES
MEET OUR TEAM

Rich Ganz - CEO
30+ years of life science industry experience

Davis Arrick - CEO
4 years of manufacturing industry experience

You-Yeon Won - CSO
10 years developing novel nanomedicine tech
ADVISORS
Arindam Bose, PhD
Purdue University, Former VP at Pfizer, Member of National Academy of Engineers
Wade Lange, MS
Purdue University, Former President/CEO Immune Works
COLLABORATORS
Krishan Raghavendran, MD
University of Michigan, Division Chief of Acute Care Surgery
Perenlei Enkhbaatar, MD, PhD
University of Texas Medical Branch, Director of Anesthesiology and Translational ICU Professor



CONTACT US
Help us make real world change. Please fill out this form below.